CBD has emerged in recent years as a potential remedy for a variety of conditions. A new study suggests that CBD may be toxic to the liver in high doses.
Worrying results
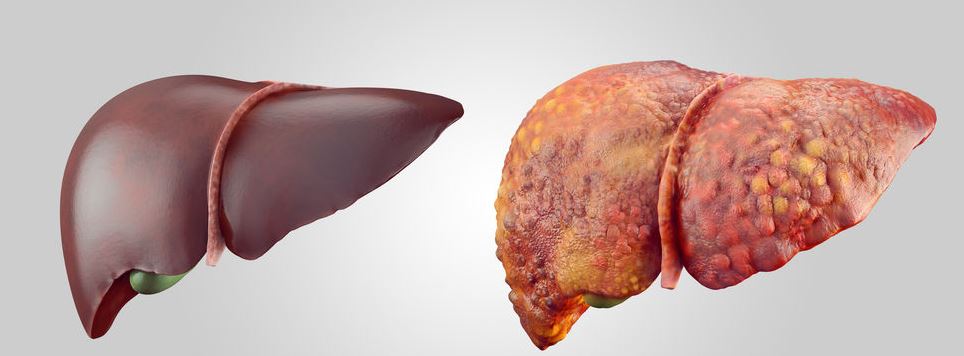 Cannabis interacts with CB1 and CB2 receptors in the endocannabinoid system. These are mainly found in the brain, but there is also a high concentration in other organs, including the liver. Taking CBD, therefore, has the potential to alter the normal functioning of the liver. It is also known to block the degradation of certain drugs by the liver and is, therefore, contraindicated in combination with certain treatments.
Cannabis interacts with CB1 and CB2 receptors in the endocannabinoid system. These are mainly found in the brain, but there is also a high concentration in other organs, including the liver. Taking CBD, therefore, has the potential to alter the normal functioning of the liver. It is also known to block the degradation of certain drugs by the liver and is, therefore, contraindicated in combination with certain treatments.
Read Also: Abusing Cannabis Increases the Risk of Testicular Cancer
Researchers from the University of Arkansas for Medical Sciences wanted to go further by conducting a study on rats to study the possible liver toxicity of CBD. To determine the doses to be administered, the researchers referred to the safety recommendations given for Epidiolex. This CBD oil which is used in the treatment of epilepsy is the first FDA-approved cannabis-based drug.
The maximum recommended daily dose for humans is 20mg/kg. This limit was then adapted for a mouse experiment. Mice were administered with higher or lower doses of CBD, some in one day (acute poisoning) and others in about ten days (progressive poisoning). They received 0, 246, 738 or 2460 mg/kg in 24 hours or daily doses of 0, 61.5, 184.5 or 615 mg/kg for 10 days.
At 2460 mg/kg, mice showed immediate signs of liver degradation: increased liver weight relative to the body, increased concentration of ALT and AST enzymes in plasma, indicating destruction of liver cells and increased levels of bilirubin, a product of red blood cell disintegration that is normally metabolized by the liver and whose accumulation indicates liver dysfunction. At 615 mg/kg per day, 75% of rats developed the liver disease after the third or fourth day.
The gene expression tables related to hepatotoxicity have shown that CBD regulates more than 50 genes, most of which are related to oxidative stress response, the pathways of lipid metabolism and enzymes of drug metabolism. “In conclusion, CBD shows clear signs of hepatotoxicity, possibly of a cholestatic nature (obstacle to the evacuation of bile). The involvement of many pathways of chemical synthesis associated with lipid and xenobiotic metabolism raises serious questions about the potential interaction of CBD with drugs and its overall safety.
In other words, CBD appears to disrupt the ability of the liver to metabolize a number of molecules, including lipids – leading to an accumulation of fat in the liver, also known as liver steatosis – and some therapeutic molecules, including antibiotics. Also, with very high doses, it appears that it can also be highly toxic, causing damage and destruction of liver cells.
Epidiolex Side Effects
The study director, Igor Koturbash, says these risks are known because they are clearly indicated in the Epidiolex package insert: Looking at the Epidiolex package insert, it is clearly stated that it can cause liver damage. It is specified that the levels of the patient’s enzymes must be monitored. In clinical studies, 5-20% of patients developed high levels of enzymes and some had to be removed from the study.
Read also: Cannabinoid Compounds May Halt Colon Cancer Growth
The liver reaction seems to depend on the patients, their condition and the dose ingested. It seems, among other things, that CBD use is contraindicated in hepatitis C: “Patients with hepatitis C who used cannabis had more liver scarring than those who did not have hepatitis C. The liver disease also progressed more. Something in the cannabis could actually be increasing fatty liver disease,” explains Dr. Hardeep Singh, a Californian gastroenterologist. These results are supported by other studies as well. However, in the case of alcohol-induced liver steatosis, CBD appears to have a positive effect.
Benefits versus Risks
For the time being, no CBD poisoning has been reported in humans and the WHO believes that the molecule is safe and without risk of abuse. In addition, most conventional drugs also damage the liver. The same applies to alcohol. This should not invalidate its medical use, but raises a number of questions about its use in consumer products and maximum exposure limits.
References
Related Articles:
Cannabis Laws: Things to Know Before Traveling with Marijuana
Why Cannabis-Derived CBD is Becoming More Popular for Health and Wellness Products
Latest CBD Laws: Everything You Need to Know About the Legality of CBD Oil
Legalization of Marijuana Has Decreased Black Market Sales
Drinking Coffee Can Reduce Liver Cancer Risk by Half Shows Queen’s University Study
Study Reveals Fat Loss Drug May Prevent Liver Disease in People Living with HIV
Immunotherapy Could Be The Key To Liver Cancer Prevention
Fibrinogen key To Liver Regeneration According To Study
FEEDBACK:


In high doses, anything is toxic to the body. Eating too many hot dogs has been proven to cause cancer. Drinking too much water has led to death. Drinking too much alcohol causes liver damage. Eating too much sugar can also be fatal. Yet, none of those have been banned. CBD was identified in the 1940s and its benefits are well documented in science. What are the benefits to alcohol?
Thank you for the interesting post! If I read this correctly, the lowest single dose the researchers gave the mice was 246 mg per kilo. This is equivalent to an 80-kg human consuming a whopping 19,680 mg of CBD.
The highest dose is ten times that: an 80-kg human would have to consume 191,000mg of pure CBD—or drink 3,800 glasses of 5% CBD oil in one day!
My worry is that this study is so fundamentally flawed that people will simply ignore the possibility of CBD causing liver damage.